  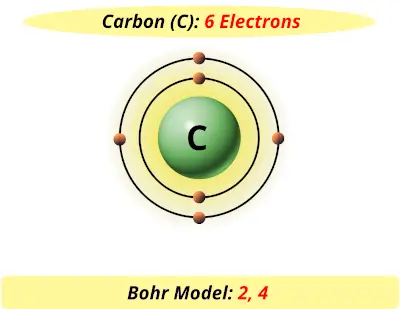
The 3rd shell can hold 18, but the elements in the first few periods only use 8 electrons. The 2nd shell can hold up to 8 electrons. Only two electrons can fit in the 1st shell. You should have 6 total electrons for Carbon. Add one at a time -starting on the right side and going counter clock-wise. 6p 6nħ Bohr Diagrams Since you have 2 electrons already drawn, you need to add 4 more. 
The first shell can only hold 2 electrons. Carbon is in the 2nd period, so it has two energy levels, or shells.Ħ Bohr Diagrams 6p 6n Add the electrons. Elements in the 2nd period have two energy levels, and so on.ĥ Bohr Diagrams 6p 6n 7) Draw the shells around the nucleus. Protons and neutrons each have a mass of 1 atomic mass unit (a.m.u.) Carbon’s atomic mass rounds to 12, so 12 – 6 protons = 6 neutrons Write 6n 6)Draw a small circle around the numbers to indicate the nucleus.Ĥ Bohr Diagrams Find out which period (row) your element is in.Įlements in the 1st period have one energy level. 6n 5) Next, figure out the number of neutrons. 6 C Carbon 12.011 Carbon is element 6, so it has 6 protons and a neutral carbon atom has 6 electronsģ Bohr Diagrams 6p 4) For carbon, write 6p where your nucleus is going to be. In a neutral atom (no net charge) it is the same as the number of protons. Presentation on theme: "How to Draw Bohr Diagrams and"- Presentation transcript:Ģ Bohr Diagrams Find your element on the periodic table.ĭetermine the number of protons, which is given by the atomic number. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
